 Particles predicted by supersymmetric theories.The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 1/1,836the mass of a proton. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. Is an electron a subatomic particle?Įlectron, lightest stable subatomic particle known. His atom however was like a solid billiard ball. John Dalton (1766-1844), a great chemist, really started the modern atomic hypothesis. These are particles that are even smaller than a single atom, and are thus called Subatomic Particles. Subatomic Particles Even though astrophysicists looks at the largest objects in the universe, you can’t understand how they work unless you also understand the smallest objects in the universe. Are subatomic particles smaller than atoms? There are three subatomic particles, which are namely electrons, protons and neutrons. What are the subatomic particles Class 8? It is 100 years since Ernest Rutherford published his results proving the existence of the proton. 
They are so small that for practical purposes they do not contribute to the mass of the atom. Electrons have a negative charge but are extremely small and have a mass only 1/1850 that of a proton or neutron. The third type of stable particle is the electron. Neutrons and protons constitute almost all of an atom’s mass. The vast majority of them are hydrogen (humans are almost entirely water, which comprises two hydrogen atoms and an oxygen). Suzanne Bell, an analytical chemist at West Virginia University, estimates that a 150-pound human body contains about 6.5 octillion (that’s 6,500,000,000,000,000,000,000,000,000) atoms. Which subatomic particle is the heaviest? Thomson was the Cavendish professor of Experimental Physics at Cambridge University and director of its Cavendish Laboratory from 1884 until 1919. Thomson, 1856-1940 see photo at American Institute of Physics) is widely recognized as the discoverer of the electron. What is the first subatomic particle discovered? If there are many of them and they are emitting some radiation, and also if we shine some radiation on then and receive back the response this will also constitute a kind of seeing. We can never see the subatomic particles directly, but can only infer from observation of such indirect effects like tracks. Or more than a vigintillionbut less than acentillion. How many subatomic particles are in the universe?ģ.28 x 10 80. The 3 subatomic particles consist of protons, electrons and neutrons. These particles are responsible for the atomic number, atomic mass and the reactions that any molecule undergoes. The structure of an atom is made up of mainly 3 subatomic particles. Electron (e-) is negatively charged particle that can occupy a volume of space (orbital) around an atomic nucleus. 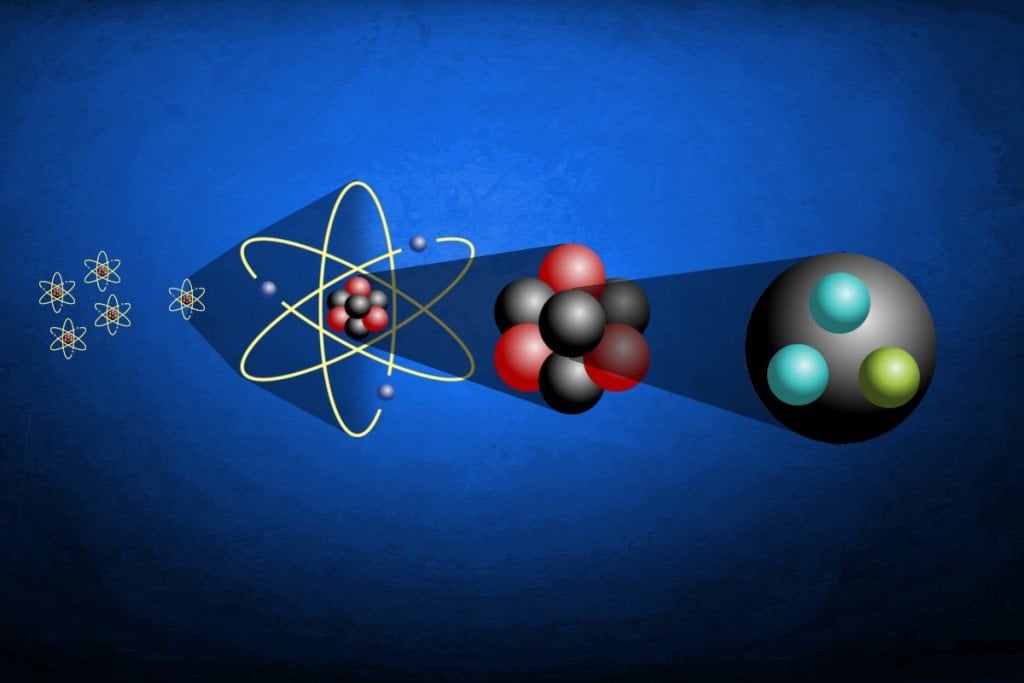
The atomic number of an element represents the number of protons in the nucleus. Proton (p+) is positively charged particle of the atomic nucleus. What are the properties of the subatomic particles? The protons have a positive electrical charge and the neutrons have no electrical charge. The nucleus contains two types of subatomic particles, protons and neutrons. Subatomic synonyms In this page you can discover 4 synonyms, antonyms, idiomatic expressions, and related words for subatomic, like: sub-atomic, fundamental-particle, quantum-theory and antimatter. Typically, an atom can be broken down into different subatomic particles, namely: protons, electrons, and neutrons. What are subatomic particles Class 11?Ī subatomic particle is nothing but a particle that is smaller than an atom in size. Subatomic particles are typically located in two places the protons and neutrons are in the nucleus at the center of the atom, whilst the electrons are in diffuse orbitals around the nucleus. number of neutrons = mass number – atomic number. To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. 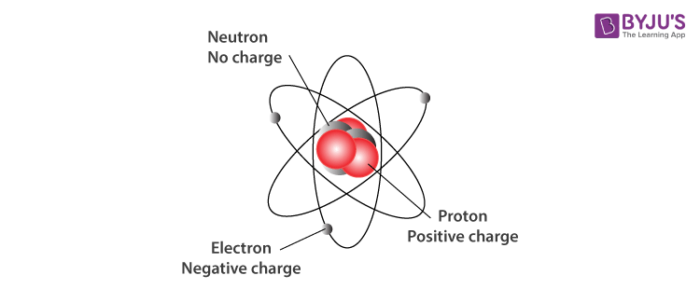 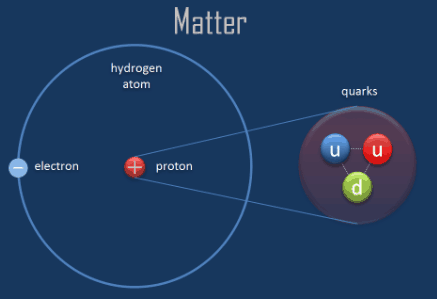
The electrons have a negative electrical charge. A third type of subatomic particle, electrons, move around the nucleus. 24 What are smaller than atoms? What does subatomic mean in physics? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
